 
The reactivity decreases up the group, making lithium the least reactive element. Francium is the most reactive alkali metal, located in the seventh row. They react with water quickly and must be stored in oil. This is why alkali metals combine with other elements in compounds. They are highly reactive and can burst into flames when exposed to air. These elements have one valence electron. The alkali metals are all elements of Group I except hydrogen. 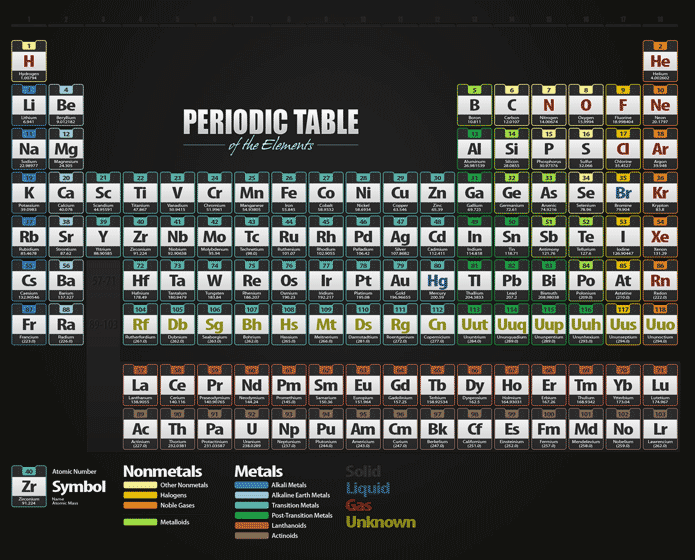
Group Numberĭ and f block metals have two valence electrons Below are the different groups of the periodic table. These electrons are either donated by, accepted from, or shared with other elements during chemical bonding. The elements in each group have the same number of valence electrons and hence, have similar chemical properties determined by the outermost electrons. According to the IUPAC system of naming groups, there are 18 groups, with the group number ranging from 1 to 18. The vertical columns of the periodic table are called groups. As we go down the period, the number of electron shells increases. In the first period, there are two elements, each having only one electron shell. The Arabic numbering system is the most widely accepted today.There are seven periods, with the period number ranging from 1 to 7. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.Halogens: - Group 17 (VIIA) - 7 valence electrons.Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons.Carbon Group or Tetrels: - Group 14 (IVA) - 4 valence electrons.Boron Group or Earth Metals: Group 13 (IIIA) - 3 valence electrons.Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons.Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons.Alkali Metals: Group 1 (IA) - 1 valence electron.Many chemists and chemistry textbooks recognize five main families:Īnother common method of categorization recognizes nine element families: However, there are different ways of categorizing elements into families. Because element properties are largely determined by the behavior of valence electrons, families and groups may be the same. Element groups, on the other hand, are collections of elements categorized according to similar properties. The characteristics of the elements in these families are determined primarily by the number of electrons in the outer energy shell. Elements are classified into families because the three main categories of elements (metals, nonmetals, and semimetals) are very broad. Element families are indicated by numbers located at the top of the periodic table.Īn element family is a set of elements sharing common properties. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
